Research
Catalysis with Halogen Bonding
Nature uses noncovalent interactions to selectively activate substrates for catalysis. The development of synthetic catalysts, on the other hand, has focused on strong covalent, dative, and hydrogen bonding. The employment of halogen bonding is underdeveloped in synthetic systems. To address this, we exploit the attractive interactions between electron-rich species and halogen bond donors. The halogen bonding catalysts we develop will have implications in pharmaceutical synthesis and organocatalysis.

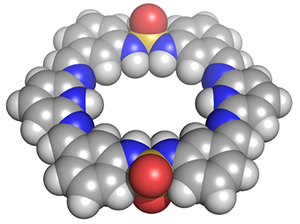
Anion Supramolecular Chemistry Utilizing Halogen Bonding
Our research group seeks to design, sythesize, and study halogen bonding supramolecular architectures that incorporate charge-diffuse anions and halides in solution and the solid state. These studies have implications in biological and environmental sensing, as well as self-assembly.
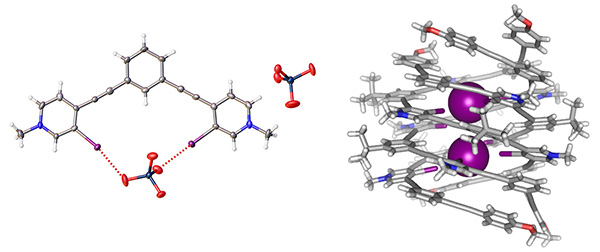
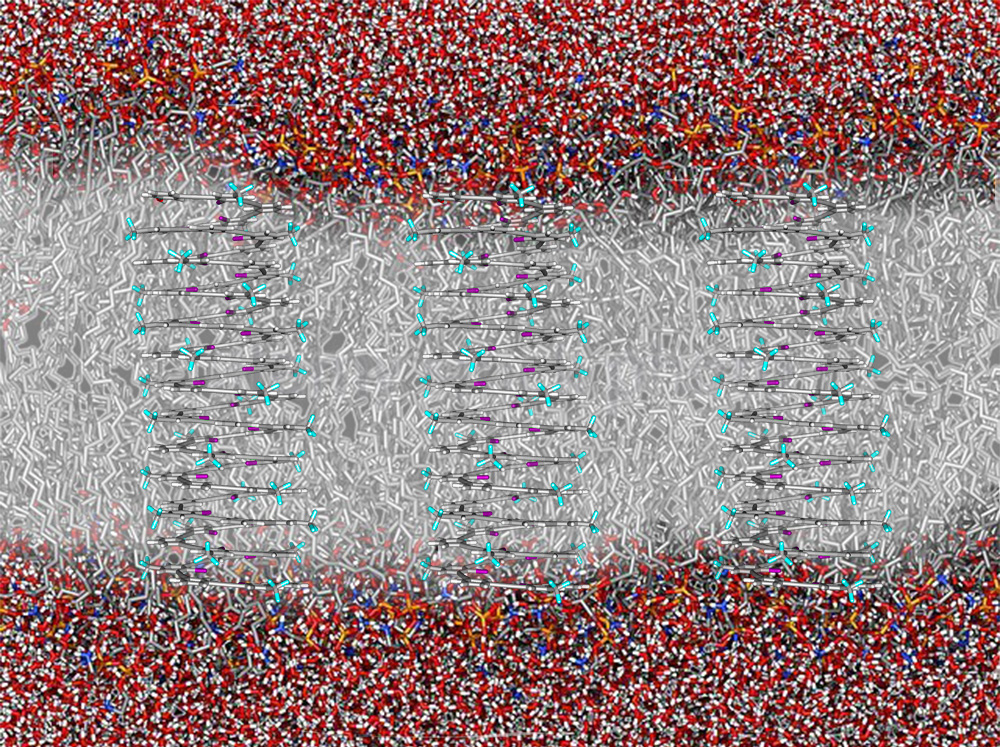
Anion-Conducting Synthetic Channels
Over the past four decades, the development of synthetic ion channels has been limited by the feasiblity of creating large and functional molecular assemblies. Tuning the amphiphilicity and size-selectivity of channel-forming foldamers, we seek to exploit our expertise in supramolecular anion self-assembly to design selective and efficient anion channels for biophysical research.
Covalent Self-Assembly of Ligands Targeting Ions with Atypical Geometries
Many anions and metals with unusual coordination geometries are biologically and/or environmentally important. Unfortunately, their unique coordination geometries are inherently difficult to target. We utilize dynamic covalent chemistry to assemble selective and efficient ligands that would be otherwise difficult to predict and prepare. Post-synthetic modification or target based synthesis will produce suitable ligands for environmental remediation of anions and metals such as UO22+.